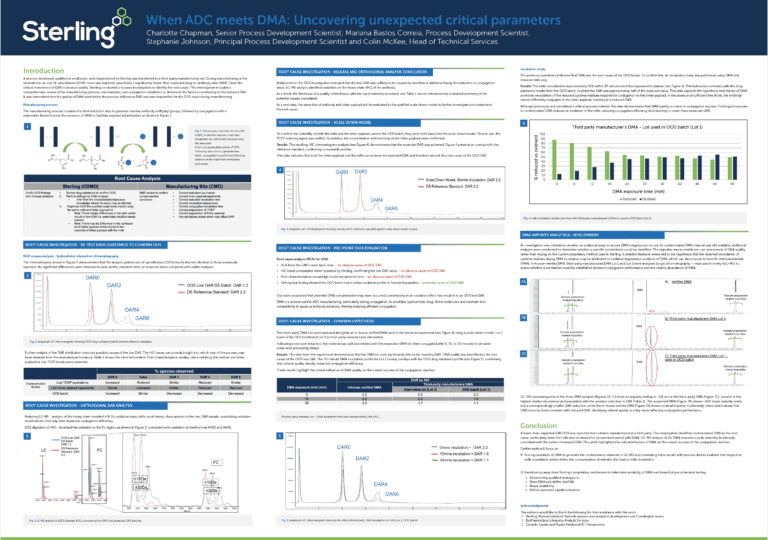
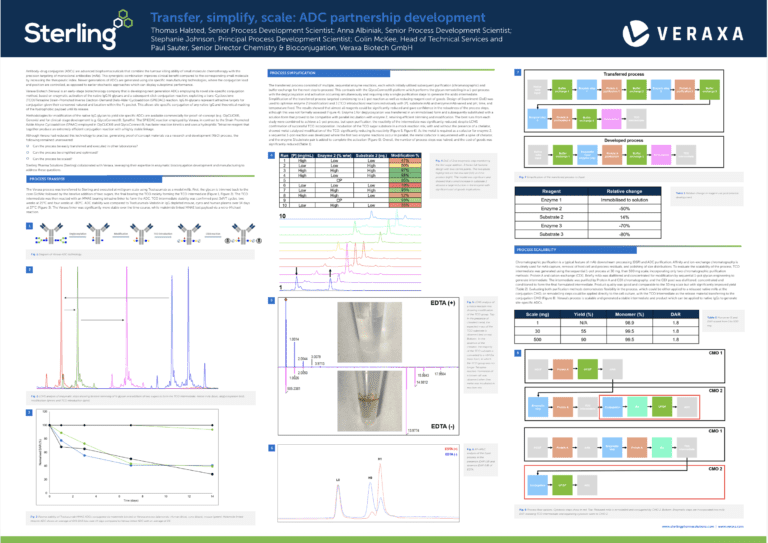
The power of peptide therapeutics: Advancements and methods in peptide drug development
Introduction: The growth of peptide therapeutics
As the pharmaceutical industry has heightened its focus on targeted therapies and personalised medicine, peptide therapeutics have emerged as a promising strategy.
While the earliest peptide therapeutics came from natural sources, limitations like low oral bioavailability and short half-lives have caused reluctance around peptide research in the past . Over the last several decades, however, advances in technology have enabled scientists to enhance peptide functionality and synthesise novel peptides for a particular purpose.
Now, the global peptide therapeutics market is valued at $43.45 billion , and this number continues to grow. With their high specificity, precise targeting capabilities, and opportunities for custom design, peptides have become a core focus for many pharmaceutical and biotechnology organisations. At Sterling, we provide the specialised expertise and equipment required to support peptide development and manufacturing at scale.
In this whitepaper, we’ll cover the advantages and core use cases for peptides in the pharmaceutical space, then compare methods for peptide drug development.
Peptide therapeutic advancements
Peptide therapeutics themselves are not a new discovery. Insulin, for example, was first used to treat diabetes in the 1920s . However, insulin and other peptide therapeutics at the time were derived from natural sources. Today, scientists are using technological advances to develop novel, targeted peptides.
Chemical synthesis of peptides first occurred in the 1950s, but peptide drugs still brought a number of challenges. As scientists began to apply strategies like the incorporation of non-natural amino acids and backbone modification, they have been able to extend half-life, improve solubility, and increase stability, making peptide therapeutics even more appealing . Today, more than 80 peptide drugs have been approved worldwide, with over 30 non-insulin peptide therapeutics entering the market since 2000 . Now, approximately 150 peptides are currently undergoing clinical development, with over 400 in preclinical studies.
Peptides are broadly used across a variety of indications. Today, as summarised by Wang et al., some of their key use cases and areas of exploration include:- Diabetes mellitus: In addition to insulin, GLP-1 receptor agonists (GLP-1RAs), including semaglutide, have gained traction in treating diabetes and obesity. They work by enhancing insulin secretion, inhibiting glucagon secretion, and slowing gastric emptying.
- Cardiovascular disease: Some food-derived peptides have shown promise in preclinical studies for inhibiting angiotensin-converting enzyme (ACE) activity and lowering blood pressure. In addition, peptides in the natriuretic peptide (NP) family have been explored in trials for heart and renal failure.
- Intestinal disease: For inflammatory bowel disease (IBD), antimicrobial and regulatory peptides, like proline-arginine-39, have shown promise in repairing the intestinal barrier and reducing inflammation. Teduglutide, a GLP-2 analogue, promotes intestinal growth and is approved in treating short bowel syndrome (SBS).
- Cancer: Peptide therapeutics are gaining traction in precision oncology due to their precise targeting capabilities. Examples include mifamurtide, which is approved for treating osteosarcoma, and carfilzomib, which is approved for multiple myeloma
The advantages of peptide therapeutics
Let’s take a closer look at some of the key advantages of peptide therapeutics, which have contributed to their growing applications in the pharmaceutical space.
Scientists can design peptides to target specific receptors, maximising therapeutic efficiency while minimising side effects. This allows for more targeted therapies and supports the development of personalised medicines.
Tumour-homing peptides, such as the arginylglycylaspartic acid (RGD) peptide, are designed to improve internalisation of nanoparticles into cancerous cells.
Naturally occurring peptides play a role in numerous physiological processes. As a result, peptide drugs can be applied to a wide range of treatment areas, and administered in a variety of ways, including injections, oral formulations, and others.
Peptides therapeutics have been developed to treat HIV, chronic pain, short bowel syndrome, and numerous other indications.
Synthesis and other biological methods for peptide development allow scientists to focus on and optimise certain properties, like stability and bioavailability. In addition, scientists can develop peptides for specific targets or pathways.
One example is CB1, a custom peptide derived from the naturally occurring Cecropin B to target lung cancer cells while avoiding healthy lung cells.
Examining methods of peptide therapeutic development
Modern peptide therapeutics are generally developed in one of two ways; through peptide synthesis, or using biological routes. In peptide synthesis, amino acids are chemically assembled to create a peptide chain, while biological routes involve cell cultivation to express and then extract the desired peptide. ... Read more
One of the most common methods of synthesis is solid-phase peptide synthesis (SPPS), which builds an amino acid sequence on a solid support. After synthesis, the product is purified to remove byproducts and isolate the active component, which is then dried using lyophilization to create the resulting peptide drug. Today, scientists can leverage automated synthesisers, such as microwave-assisted peptide synthesisers, to build peptides with greater efficiency than ever. Purification is then necessary to remove any byproducts and isolate the target peptide.
Both peptide synthesis and biological approaches to customisation can be beneficial in certain use cases. While biological routes deliver advantages like post-translational modifications and can support potentially more complex structures, chemical peptide synthesis can enable greater cost-effectiveness, more precise control, higher stability, and faster development.Read less
Selecting the most appropriate method
When discerning whether to leverage peptide synthesis or biological methods in their programmes, scientists should consider several factors. Here are some of the potential advantages each approach can deliver:
• Precise control over amino acid sequence
• Lower setup costs
• Rapid development
• Higher stability
• Access to non-natural peptides
• Support for complex structures
• Potential for post-translational modifications
• Higher specificity
• Environmental friendlines
What’s next in peptide drug development?
As the peptide therapeutics market continues to expand, with an expected 10.77% CAGR through 2030 , several new peptide formats and delivery methods are emerging. Next-generation peptide therapeutics aim to overcome some of the shortcomings associated with traditional peptides, such as stability and permeability.
Let’s explore some novel peptide formats that are gaining traction in pharmaceutical development and manufacturing.
Stapled peptides
Stapled peptides use a stable, alpha-helix structure to support greater membrane permeability and stability. Several therapeutic stapled peptides have reached clinical trials, but none are approved today.
Cyclic and bicyclic peptides
Cyclic and bicyclic peptides are being explored for their strong stability and bioavailability. They show promise in targeting challenging protein-protein interactions, and bicyclic peptides in clinical trials today largely focus on oncology and antimicrobial applications.
Peptide-drug conjugates
Similar to antibody-drug conjugates (ADCs), Peptide-drug conjugates (PDCs) combine peptides, linkers, and toxic payloads to target specific cells. They are small relative to ADCs, supporting greater tumour penetrability. Two PDCs have been clinically approved to date, with more in preclinical development.
Maximising peptide potential with the right partner
Now is a more exciting time than ever for peptide therapeutics, with over 170 peptide drugs in clinical development today and even more in the preclinical stage. As peptides remain a core area of research and progress in the life sciences space, pharmaceutical and biotechnology organisations require a partner with extensive expertise. ... Read more
Sterling offers end-to-end support in peptide drug development and manufacturing, helping scientific organisations scale complex peptide therapeutics with precision and efficacy.
Our Cork, Ireland facility offers more than 20 years of expertise in peptide synthesis, analysis, manufacturing and purification, and we are authorised to supply peptides to all markets. Our facilities include a dedicated laboratory for peptide analysis and a multipurpose plant with a peptide synthesiser and stirred reactors. In addition, our purification plant consists of multiple chromatography columns, bioburden controls and cleanroom lyophilization capability, allowing us to support a full range of peptide synthesis requirements. We combine powerful capabilities with close collaboration to empower our customers and support industry-wide innovation.Read less
FAQs
Peptide therapeutics are composed of short amino acid chains, typically with fewer than 50 amino acids. They are designed to mimic or facilitate biological processes, enabling precise targeting capabilities with strong biological activity.
Peptide therapeutics may be created with naturally occurring peptides, or developed synthetically.
Peptide therapeutics fall somewhere between small molecule and biologic drugs. Due to their precise targeting capabilities, they can treat a variety of indications safely and effectively, and they are generally well-tolerated.
Peptide therapeutics show strong potential in a range of therapeutic areas, with numerous peptides approved today to treat diabetes, cancer, gastrointestinal disease, and more.
80+ peptide therapeutics are currently approved, and more than 150 are in clinical trials today.
Peptide drug development brings a range of challenges and considerations that must remain a focus throughout the development and manufacturing process.
Some challenges relate to their pharmacokinetic properties, such as limited stability and permeability. Next-generation peptide therapeutics like stapled peptides and PDCs aim to address these challenges.
Another challenge is their manufacturing complexity. Developing and purifying peptides generally requires specialised equipment and expertise, and not all outsourced manufacturing partners offer capabilities in this area. Furthermore, longer peptide chains can bring additional challenges surrounding purification.
A partner experienced in peptide development and manufacturing can help you navigate these challenges and deliver tailored, high-quality peptide therapeutics efficiently.
Next-generation peptide therapeutics aim to enhance some of the shortcomings of standard peptides, engineering them for greater stability, bioavailability, and other properties. Next-gen peptide drug development techniques include stapled peptides, cyclic peptides, and peptide-drug conjugates.
At Sterling, we deliver extensive expertise in peptide development, manufacturing, purification, and analysis. We are experienced in both solid- and liquid-phase peptide synthesis. Our work is supported by dedicated analytical laboratories for peptide analysis and release, peptide synthesisers with up to 170L capacity, and one of the world’s largest HPLC columns for peptide purification at scale.