Transfer, simplify, scale: ADC partnership development
Antibody-drug conjugates (ADCs) are advanced biopharmaceuticals that combine the tumour killing ability of small molecule chemotherapy with the precision targeting of monoclonal antibodies (mAb). This synergistic combination improves clinical benefit compared to the corresponding small molecule by increasing the therapeutic index. Newer generations of ADCs are generated using site specific manufacturing technologies, where the conjugation load and position are controlled, as opposed to earlier stochastic approaches which can display suboptimal performance.
Veraxa Biotech (Veraxa) is an early-stage biotechnology company that is developing next generation ADCs employing its novel site-specific conjugation method, based on enzymatic activation of the native IgG N-glycans and a subsequent click conjugation reaction, exploiting a trans-Cyclooctene (TCO)/Tetrazine Strain-Promoted Inverse Electron-Demand Diels-Alder Cycloaddition (SPIEDAC) reaction. IgG N-glycans represent attractive targets for conjugation given their conserved natured and location within the Fc pocket. This allows site specific conjugation of any native IgG and theoretical masking of the hydrophobic payload until its release.
Methodologies for modification of the native IgG glycan to yield site specific ADCs are available commercially for proof-of-concept (e.g. GlyCLICK®, Genovis) and for clinical-stage development (e.g. GlycoConnect®, Syna x). The SPIEDAC reaction employed by Veraxa, in contrast to the Strain Promoted Azide Alkyne Cycloaddition (SPAAC) employed in GlyCLICK® and GlycoConnect®, has faster reaction kinetics and uses a hydrophilic Tetrazine reagent that together produce an extremely efficient conjugation reaction with a highly stable linkage.
Although Veraxa had reduced this technology to practise, generating proof of concept materials via a research and development (R&D) process, the following remained unanswered:
- Can the process be easily transferred and executed in other laboratories?
- Can the process be simplified and optimised?
- Can the process be scaled?
Sterling Pharma Solutions (Sterling) collaborated with Veraxa, leveraging their expertise in enzymatic bioconjugation development and manufacturing to address these questions.
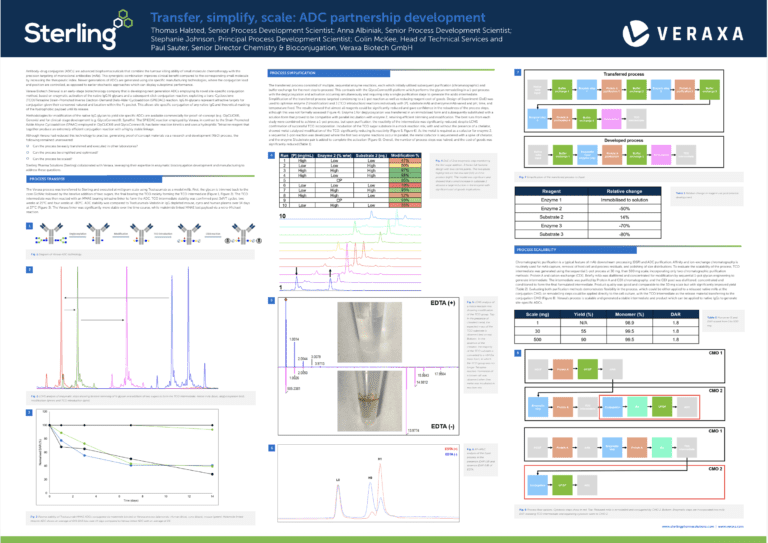
Process transfer
The Veraxa process was transferred to Sterling and executed at milligram scale using Trastuzumab as a model mAb. First, the glycan is trimmed back to the core GlcNAc followed by the iterative addition of two sugars, the final bearing the TCO moiety forming the TCO intermediate (Figure 1, Figure 2). The TCO intermediate was then reacted with an MMAE bearing tetrazine linker to form the ADC. TCO intermediate stability was confirmed post 3xF/T cycles, two weeks at 25°C and four weeks at -80°C. ADC stability was compared to Trastuzumab-Vedotin in IgG depleted mouse, cyno and human plasma over 14 days at 37°C (Figure 3). The Veraxa linker was significantly more stable over the time course, while maleimide linked MMAE lost payload via a retro-Michael reaction.
Process simplification
The transferred process consisted of multiple, sequential enzyme reactions, each which initially utilised subsequent purification (chromatographic) and buffer exchange for the next step to proceed. This contrasts with the GlycoConnect® platform which performs the glycan remodelling in a 1-pot process with the deglycosylation and activation occurring simultaneously and requiring only a single purification stage to generate the azido intermediate. Simplification of the transferred process targeted condensing to a 1-pot reaction as well as reducing reagent cost of goods. Design of Experiment (DoE) was used to optimise enzyme 2 (modification) and 3 (TCO introduction) reactions individually with [P], substrate:mAb and enzyme:mAb varied and pH, time, and temperature fixed. The results showed that almost all reagents could be significantly reduced and gave confidence in the robustness of the process steps, although this was not formally assessed (Figure 4). Enzyme 1 for deglycosylation was transferred in an immobilised form and subsequently substituted with a solution form that proved to be compatible with parallel incubation with enzyme 2, retaining efficient trimming and modification. The best runs from each study were combined to achieve a 1-pot process, but upon purification, the reactivity of the intermediate was significantly reduced, despite LCMS confirmation of successful TCO incorporation. Incubation of the TCO sugar substrate in a mock reaction mix, with and without the presence of a chelator, showed metal catalysed modification of the TCO, significantly reducing its reactivity (Figure 5, Figure 6). As the metal is required as a cofactor for enzyme 2, a sequential 1-pot reaction was developed where the first two enzyme reactions occur in parallel, the metal cofactor is sequestered with a spike of chelator, and the enzyme 3/substrate pair is added to complete the activation (Figure 6). Overall, the number of process steps was halved, and the cost of goods was significantly reduced (Table 1).
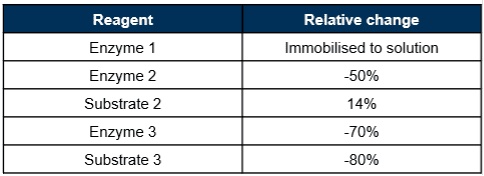
Table 1. Relative change in reagent use post process development.
Process scalability
Chromatographic purification is a typical feature of mAb downstream processing (DSP) and ADC purification. A nity and ion-exchange chromatography is routinely used for mAb capture, removal of host cell and process residuals, and polishing of size distributions. To evaluate the scalability of the process, TCO intermediate was generated using the sequential 1-pot process at 30 mg, then 500 mg scale, incorporating only two chromatographic purification methods: Protein A and cation-exchange (CEX). Briefly mAb was diafiltered and concentrated for modification by sequential 1-pot glycan engineering to generate intermediate. The intermediate was purified by Protein A and CEX chromatography, and the CEX pool was diafiltered, concentrated and conditioned to form the final formulated intermediate. Product quality was good and comparable to the 30 mg scale but with significantly improved yield (Table 2). Evaluating both purification methods demonstrates flexibility in the process, which could be either applied to a released native mAb at the conjugation CMO, or remodelling steps could be applied directly to the cell culture, with the TCO intermediate as the release material transferring to the conjugation CMO (Figure 8). Veraxa’s process is scalable and generated a stable intermediate and product which can be applied to native IgGs to generate site-specific ADCs.
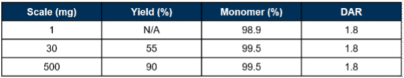
Table 2. Monomer % and DAR scaled from 1 to 500 mg.
Figure 8. Process flow options. Cytotoxic steps show in red. Click for Figure 8a. Released mAb is remodelled and conjugated by CMO 2. Click for Figure 8b. Enzymatic steps are incorporated into mAb DSP, releasing TCO intermediate and separating cytotoxic work to CMO 2.