Scaling ring-closing metathesis in API development: Challenges and success factors
As active pharmaceutical ingredients (APIs) continue to increase in structural complexity, process chemistry teams are increasingly challenged to construct demanding molecular architectures during both development and commercial manufacturing. Cyclic motifs are particularly prevalent in modern drug design, where they play an important role in enhancing molecular stability and membrane permeability.
For synthetic routes involving difficult-to-form ring systems, ring-closing metathesis (RCM) has emerged as a powerful and versatile strategy. RCM is a subtype of olefin metathesis in which two alkene groups undergo catalytic exchange to form a new carbon–carbon double bond. This reaction enables efficient access to a wide range of ring sizes and substitution patterns, often under relatively mild reaction conditions.
Olefin metathesis has become a foundational transformation in synthetic chemistry due to its efficiency, functional group tolerance and broad applicability across diverse substrates and reaction environments. Its significance was formally recognised with the 2005 Nobel Prize in Chemistry, awarded to Yves Chauvin, Robert H. Grubbs and Richard R. Schrock for their pioneering contributions to the mechanistic understanding and catalyst development of metathesis reactions. Schrock first developed efficient catalysts for metathesis, while Grubbs later introduced ruthenium-based catalysts that offered improved operational robustness, including greater stability and tolerance to air and moisture.
Despite its widespread use in academic research and medicinal chemistry, RCM has historically seen more limited adoption in large-scale API manufacturing, as the translation from laboratory to production scale can introduce several technical and operational challenges. However, when these challenges are effectively addressed through advanced process development, specialised equipment and close cross-functional collaboration, RCM can serve as a highly effective strategy for the scalable synthesis of cyclic APIs.
Why ring-closing metathesis is valuable in pharmaceutical development
 Ring-closing metathesis offers several key advantages that make it a valuable strategy in pharmaceutical development.
Ring-closing metathesis offers several key advantages that make it a valuable strategy in pharmaceutical development.
Efficient ring formation
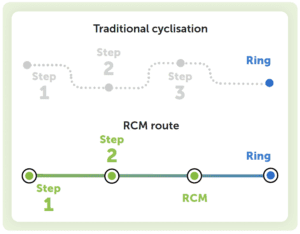
One of the most significant advantages of RCM is its ability to efficiently form cyclic structures that are difficult to construct with conventional intramolecular cyclisation or other methods.
While traditional cyclisation strategies may suffer from low yields, RCM enables the direct formation of a carbon–carbon bond. This can greatly simplify the synthesis of cyclic molecules that would otherwise require more complex strategies.
Flexibility across structural types
RCM reactions are valuable for their compatibility with a broad range of substrates. Furthermore, ruthenium catalysts can tolerate a variety of functional groups, supporting greater applicability across different reactions and structures.
Ability to streamline complex routes
In addition to facilitating difficult ring formations, RCM can help to simplify complex synthetic routes. Some ring formation strategies require multiple functional group transformations, while RCM, by contrast, can achieve the desired structure in a single transformation. This reduces the overall number of process steps, which often translates to shorter development timelines and reduced costs.
Controlling process variables during RCM scale-up
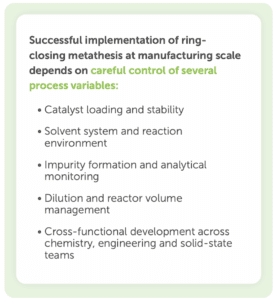
To take advantage of the benefits of ring-closing metathesis, its implementation at larger scales requires careful management of several technical considerations. Targeted process development, analytical control strategies and cross-functional expertise are required to successfully apply RCM to projects. The following highlight some typical challenges and how to overcome them.
1. Impurity formation
Grubbs catalysts remain active even after reaction completion, and so residual catalyst activity can be a challenge. If the catalyst is not properly quenched or removed, unwanted impurities or byproducts may form. Additional impurities can complicate purification and impact end product quality if not carefully controlled.
Addressing impurities requires an integrated development approach that treats catalyst control, in-process analytics and purification design as interconnected elements rather than separate workstreams.
2. Limited solvent selection
Catalyst stability at high dilution may depend heavily on solvent selection. In many cases, solvents like dichloromethane (DCM) provide optimal reaction performance, but can introduce added environmental, safety, and waste treatment considerations during scale-up.
Careful solvent screening and process optimisation can identify suitable alternatives that balance performance with environmental and safety considerations.
3. Solid state considerations
Beyond reaction optimisation, solid state behaviour can also influence the success of RCM processes. Thorough solid state evaluations are necessary to identify the most stable polymorphic form for manufacturing. Futhermore, care must be taken to avoid inadvertently isolating less soluble hydrated forms as this can affect downstream processing.
Solid state characterisation must be integrated into process development from the project’s outset rather than waiting until reaction conditions are fixed.
4. Costly catalysts
Grubbs catalysts and other specialised catalysts used in RCM tend to be relatively expensive. While this poses less of a concern at the lab scale, when these catalysts are used in small volumes, costs can quickly add up when applying this chemistry at the manufacturing scale.
While careful catalyst loading optimisation and recovery strategies can help to mitigate cost concerns, it remains an important consideration.
Parallel development of catalyst optimisation and metal removal strategies are required early on to avoid late-stage constraints.
High dilution requirements
RCM reactions often require relatively high dilution to minimise intermolecular reactions. Maintaining appropriate dilution helps favour intramolecular cyclisation, although this can introduce practical challenges at scale. Larger solvent volumes can reduce batch size and thereby increase the number of batches required to meet production demands. Furthermore, higher solvent usage can increase costs and bring additional considerations around solvent recovery and waste management.
Process optimisation and considered reactor design help balance dilution requirements with manufacturing efficiency at scale.
Scaling ring-closing metathesis: Keys to success
Although ring-closing metathesis presents several complexities when it comes to scale-up, robust process development and cross-functional expertise make efficient and high-quality manufacturing achievable.
Robust early phase process development
Comprehensive early-phase process development is essential when incorporating RCM into a synthetic route. Beyond identifying reaction conditions that work, it involves building a quantitative understanding of the reaction and systematically mapping how variables like solvent selection, dilution and catalyst behaviour ultimately impact yield, impurity profile and catalyst consumption. This enables chemists to define a reliable operating window that ensures robustness and minimises risk before progressing to larger-scale manufacturing.
Quality starting materials and analytical support
As catalysts can be sensitive, the success of RCM reactions can rely heavily on the quality of the starting materials and reagents used. In addition, advanced analytical techniques are crucial for monitoring reaction progress and identifying impurities. Methods like liquid chromatography—mass spectrometry (LC-MS) play an important role in evaluating reaction behaviour, tracking potential side reactions and ensuring effective control of impurity formation.
In addition, early solid state screening and characterisation play a key role in ensuring consistent isolation of the desired polymorphic form and minimising downstream risk.
Cross-functional collaboration
Successfully scaling a process involving sensitive catalysts and complex transformations requires a concerted effort across multiple scientific disciplines. When teams work in silos and sequentially hand off project steps, challenges and interactions that may influence downstream success can easily be missed Instead, process chemists, analytical chemists, solid state specialists and engineering teams must work together from the outset to assess reaction performance, manage impurities, design effective processes and align on manufacturing requirements.
Expertise in complex chemistry
Projects involving RCM require support from teams experienced in complex, multi-step syntheses. Handling sensitive catalysts, managing high dilution requirements and controlling impurity formation demand specialised expertise at both the lab and plant scale. Involving expert engineering teams early on is crucial for designing processes that will translate effectively to a larger scale while accommodating RCM reactions’ specialised requirements. Organisations with extensive experience across these areas are better positioned to successfully translate promising synthetic strategies into robust and reliable manufacturing processes, ensuring effective management of catalyst behaviour, solvent systems, dilution requirements and impurity control at scale.
Conclusion
 As pharmaceutical molecules continue to increase in structural complexity, synthetic strategies capable of efficiently constructing challenging architectures are becoming increasingly important in API development. Ring-closing metathesis represents a powerful tool for enabling the formation of complex cyclic structures that may otherwise require longer or less efficient synthetic routes.
As pharmaceutical molecules continue to increase in structural complexity, synthetic strategies capable of efficiently constructing challenging architectures are becoming increasingly important in API development. Ring-closing metathesis represents a powerful tool for enabling the formation of complex cyclic structures that may otherwise require longer or less efficient synthetic routes.
The successful implementation of ring-closing metathesis at scale requires careful management of several technical considerations. Addressing these factors through robust early-phase process development, strong analytical support and coordinated cross-functional expertise is critical to ensuring that promising laboratory reactions can be translated into reliable manufacturing processes.
At Sterling, multidisciplinary teams across our global network collaborate to support the development and manufacture for complex synthetic processes. By combining expertise in process chemistry, analytical development, solid-state science and manufacturing engineering across our international sites, we help customers successfully translate advanced synthetic strategies into robust and scalable commercial processes.