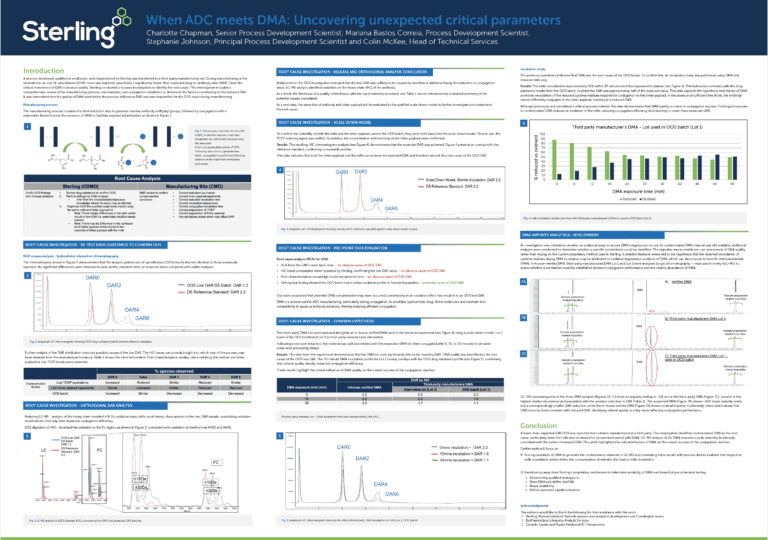
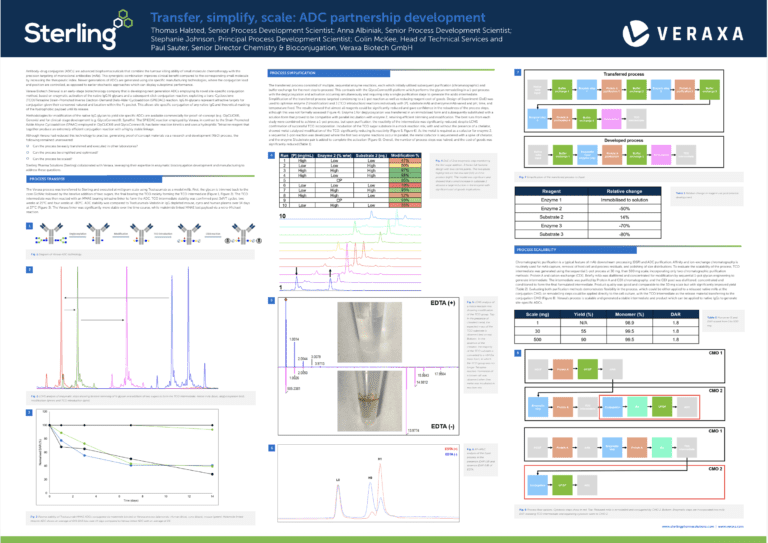
What are APIs in pharma?
In the pharmaceutical industry, active pharmaceutical ingredients, or APIs, refer to the essential components of drug products that are responsible for producing a desired therapeutic effect. In other words, an API is the therapeutically active component of a drug product that treats or prevents a specific disease or symptom. Examples of APIs include acetylsalicylic acid in aspirin or acetaminophen in Tylenol and other pain relievers.
Key takeaways:
- APIs are the active components of drug products
- They can be synthetic, natural, or high potency
- The manufacturing process includes route scouting, scale-up, GMP production, and testing
- Regulatory bodies like the FDA, EMA, and MHRA oversee API quality
APIs are a type of drug substance, though the two terms can be used interchangeably. They are distinct from excipients, or inactive ingredients, which include things like binders, fillers, coatings and more. While excipients do not exert pharmacological effects themselves, they play critical roles in ensuring drug stability, safety and efficacy. Together, APIs and excipients form the final drug product, with each component optimised to work in synergy.
Given that APIs are fundamental to how small molecule drugs work, a robust approach to API development and manufacturing is central to the success of any pharmaceutical programme. Let’s take a closer look at the importance of APIs in drug formulation, then dive into the different types of APIs and an overview of the API manufacturing process.
Why are APIs important in pharmaceutical development?
The API defines the pharmacological effect of a final drug product. Its structure-activity relationship, physiochemical properties, and purity profile influence potency, selectivity, bioavailability and other important factors.
Understanding how small molecule drugs work and interact with biological systems is critical to informing high-stakes decisions throughout the API development process. Each decision impacts not only process efficiency and safety, but also quality, purity, stability and efficacy of the final API.
Effective decision-making requires a combination of skillsets and areas of expertise, including:
- Analytical chemistry to evaluate impurity profiles, stability, and degradation pathways using techniques like HPLC, LC-MS, NMR and others
- Solid state chemistry to identify and optimise the most appropriate solid form of an API and enhance stability, solubility or bioavailability
- Process chemistry and engineering to develop scalable, efficient, and economically viable synthetic routes that maximise yield and minimise impurities
- Hazardous chemistry and containment where necessary to safely handle hazardous or highly potent APIs
Given the critical impact of API development and manufacturing on patient safety and therapeutic efficacy, a wide range of regulatory frameworks and governing bodies oversee the process to ensure that every active pharmaceutical ingredient adheres to the highest standards of quality and safety. These include:
- Good Manufacturing Practice (GMP): Refers to a universally applied quality system to guide manufacturing for APIs and drug products
- International Council for Harmonisation (ICH) Q7: Defines harmonised GMP guidelines for API manufacturing
- Medicines and Healthcare Products Regulatory Agency (MHRA): Enforces GMP compliance in the UK
- US Food & Drug Administration (FDA): Enforces cGMP compliance in the US
- European Medicines Agency (EMA): Enforces GMP compliance in Europe
Types of APIs in the pharmaceutical industry
Active pharmaceutical ingredients are broadly classified into two key categories: synthetic APIs and natural APIs. Let’s take a closer look at the distinction between the two.
Synthetic APIs are developed within a chemical process, typically a multi-step organic reaction. These types of APIs are more common due to their versatility, as well as the control afforded over the development and manufacturing process. This gives scientists the ability to control the structure of the molecule, thus controlling how the small molecule drug works and interacts with the body. Examples of synthetic APIs in pharma include ibuprofen and acetaminophen.
Natural APIs are derived from natural sources, like plants and microorganisms. They are extracted and purified to be used in final drug products. Common examples of natural APIs include cannabidiol (CBD), which comes from the cannabis plant, and morphine, which comes from poppies.
Another category of API in pharma, which is most commonly synthetic, is high potency APIs (HPAPIs). HPAPIs have grown in momentum because of their ability to deliver high levels of potency at low concentrations, and they are characterised by an occupational exposure limit (OEL) of 10 ≤10 µg/m³ or less. These types of APIs have become common in oncology drugs and other targeted therapies, but safely producing them requires thorough containment measures and safety protocols.
A look at the API manufacturing process
API manufacturing is a complex, multi-step process that requires careful monitoring and extensive cross-functional collaboration at every stage. Here is an overview of a typical API manufacturing process, though requirements and stages may vary based on specific project considerations.
- Route scouting and process development: Scientists identify a scalable synthetic pathway to produce the desired API, then optimise the process for reproducibility, safety and yield
- Purification and isolation: The API is purified and isolated in line with the required purity standards. Common purification methods include crystallisation and distillation
- Scale-up: The process moves from the lab scale to a pilot plant to assess scalability and equipment compatibility
- GMP manufacturing: The drug substance is manufactured in the plant under GMP conditions, with thorough in-process controls and quality assurance protocols
- Analytical and stability testing: Scientists test the drug substance for purity, stability, potency and other important variables using validated analytical methods, and monitor the reaction on an ongoing basis to ensure robustness of the process
- Documentation and regulatory submission: Documentation that affirms manufacturing quality and product safety is compiled to support regulatory filings
At Sterling, our expert team can support the full API lifecycle, from the lab, to clinical manufacturing, to full-scale commercial manufacturing. Our experience spans high potency API development, hazard analysis, analytical services, solid state chemistry, CMC and regulatory support and more, enabling us to help our customers produce effective and high-quality APIs no matter their specific project requirements.
If you’re interested in learning more about how Sterling supports the API manufacturing process, visit our webpage or contact an expert.