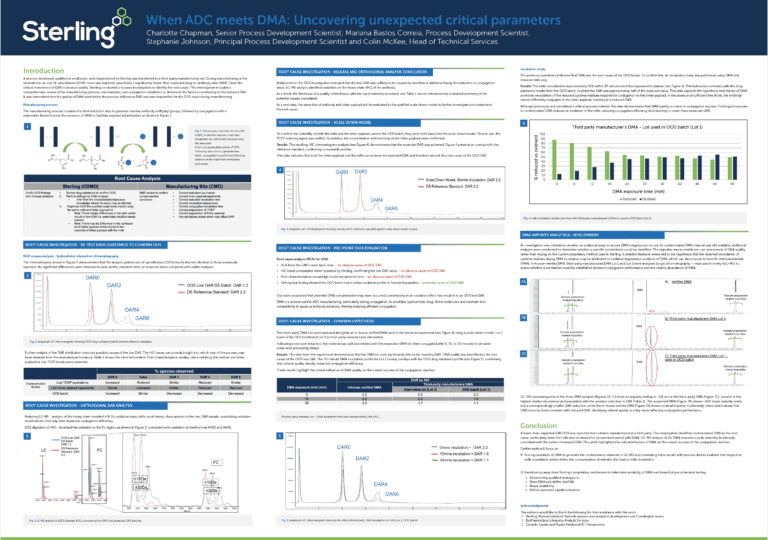
Reduced subunit analysis of a cysteine linked antibody drug conjugate
Subunit analysis of Brentuximab Vedotin using a SCIEX X500B Mass Spectrometer
Antibody-drug conjugates (ADCs) are an emerging class of biopharmaceutical combining the specific targeting nature of monoclonal antibodies (mAbs) with highly cytotoxic payloads. Currently eleven ADCs have been approved for use and there are many more in clinical trials. ADCs present unique analytical challenges due to the inherent heterogeneity of the mAb scaffolds combined with varying numbers of conjugated drug. Drug-antibody-ratio (DAR)for ADCs is a critical quality attribute (CQA) affecting the efficacy and safety of the final product. Conjugation of the toxic drug to the antibody can be done through various kinds of chemical linker reacting to different amino acids.
Here we outline an analysis strategy to confirm the DAR of an ADC with a monomethyl auristatin E (MMAE) payload bound to cysteine residues of a CD30 targeting mAb with a cathepsincleavable linker. All data was collected on a SCIEX X500B Mass Spectrometer with an EXION HPLC using a bioZen Intact XBC8 column (3.6 µm, 50 x 2.1 mm) with a 10 minute gradient of water and acetonitrile (0.1% formic acid).
Chromatographic separation of most of the species is shown in Figure 1. This ensures minimal spectral overlap leading to improved accuracy in calculating intact molecular weight. Reconstructions are shown in Figure 2 for L0 and L1 and Figure 3 for H0,1, 2, and 3. DAR can be calculated for each chain individually through reconstructed peak area, with heavy chain peaks including assigned glycans G0F, G1F, and G2F with the presence of C-terminal lysine clipping and N-terminal pyroglutamate formation. The DAR for the heavy chain was calculated at 1.44 and the light chain at 0.53 giving a total of 3.94, hydrophobic interaction chromatography was conducted on the same sample which gave a value of 4.03 and published data states a DAR of 4.
Figure 1: TIC of all chains eluting. Separation of most conjugated species is possible though H0 and L1 eluting together. See more here.
Figure 2: Reconstructed spectra of brentuximab vedotin light chain. The DAR of the light chain was calculated as 0.53 with some free linker present. See more here.
Figure 3: Reconstructed spectra of brentuximab vedotin heavy chain. The DAR of the heavy chain was calculated as 1.44. See more here.
Related Content

ADCs

ADC Analytical Services