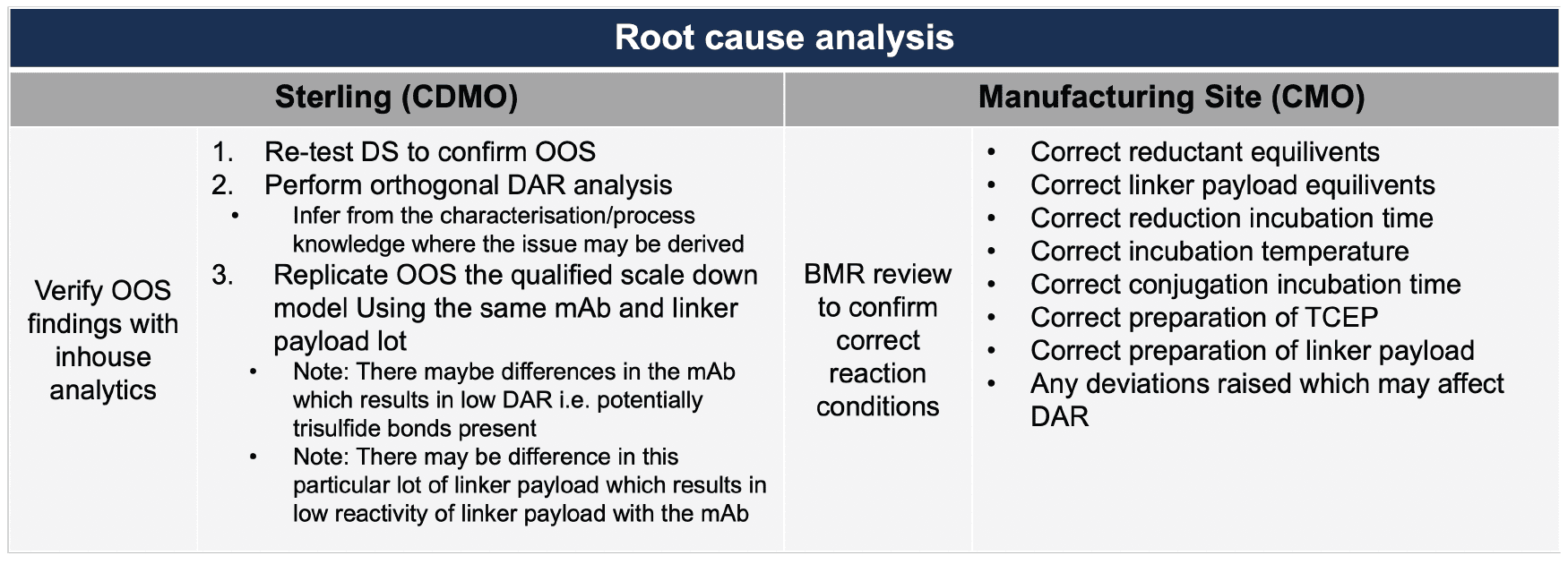
When ADC meets DMA: Uncovering unexpected critical parameters
A process developed, qualified at small scale, and characterised by Sterling was transferred to a third-party manufacturing site. During manufacturing at the external site, an out-of-specification (OOS) result was reported, specifically a significantly lower-than-expected drug-to-antibody ratio (DAR). Given the critical importance of DAR to product quality, Sterling conducted a focused investigation to identify the root cause. This investigation included a comprehensive review of the manufacturing process, raw materials, and conjugation conditions to determine the factors contributing to the reduced DAR.
Manufacturing process
The manufacturing process consists of a thiol reduction step to generate reactive antibody sulfhydryl groups, followed by conjugation with a maleimide-linked toxin in the presence of DMA to facilitate payload solubilisation, as shown in Figure 1.
Root cause investigation – re-rest drug substance to confirm OOS
DAR release analysis – hydrophobic interaction chromatography
The chromatogram shown in Figure 2 demonstrates that the analysis yielded out-of-specification (OOS) results that are identical to those previously reported. No significant differences were observed in peak profile, retention time, or response when compared with earlier analyses.
Further analysis of the DAR distribution ruled out possible causes of the low DAR. The HIC traces can provide insight into which step of the process may have deviated from the expected performance. Table 2 shows the trend information from characterisation studies, demonstrating that neither low linker payload or low TCEP trends were observed.
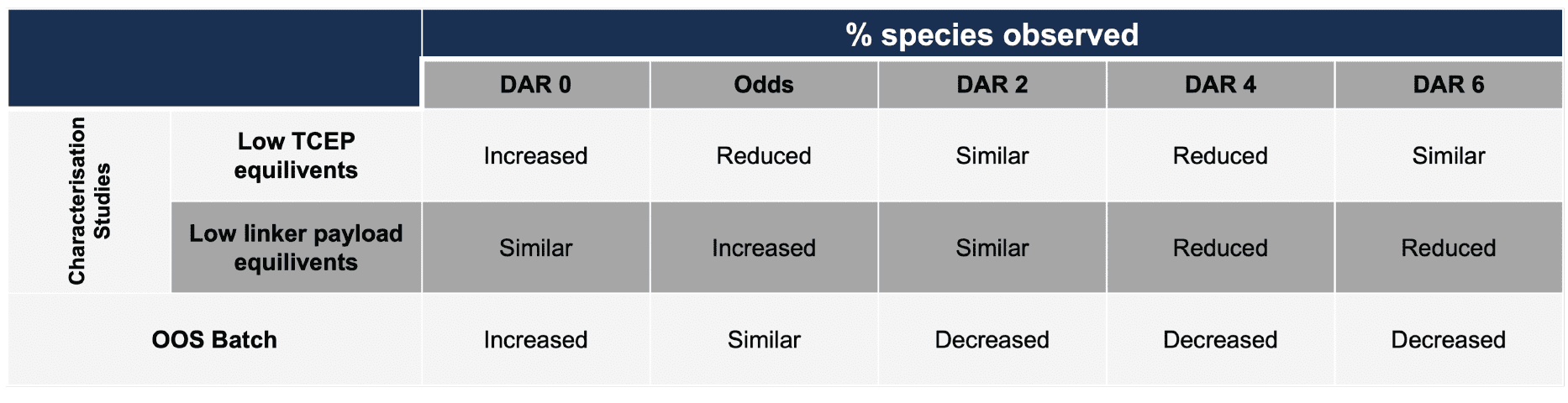
Table 2
Root cause investigation – orthogonal DAR analysis
Reducing LC-MS – analysis of the heavy chain revealed +16 Da oxidation mass shifts on all heavy-chain species in the low-DAR sample, postulating oxidative modifications that may have impacted conjugation effciency.
IDES digestion LC-MS – localised the oxidation to the Fc region, as shown in Figure 3, consistent with oxidation of methionine’s M255 and M431.
Root cause investigation – release and orthogonal analysis conclusion
Analytics from the OOS investigation indicated that the low DAR was unlikely to be caused by insuffcient additions during the reduction or conjugation steps. LC–MS analysis identified oxidation on the heavy chain (HC) of the antibody.
As a result, the likelihood of a quality-related issue with the input materials increased, and Table 1 can be referenced for a detailed summary of the potential causes considered.
As a next step, the same lots of antibody and linker payload will be evaluated in the qualified scale-down model to further investigate and understand the root cause.
Root cause investigation – scale down model
To confirm the suitability of both the mAb and the linker payload used in the OOS batch, they were both input into the scale-down model. Prior to use, the TCEP reducing agent was verified. In addition, the concentration and reactivity of the linker payload were confirmed.
Results: The resulting HIC chromatogram analysis (see Figure 4) demonstrates that the expected DAR was achieved. Figure 4 presents an overlay with the reference standard, confirming comparable profiles.
This data indicates that both the linker payload and the mAb can achieve the expected DAR and therefore are not the root cause of the OOS DAR.
Root cause investigation – mid point data evaluation
Root cause analysis (RCA) for OOS:
- RCA from the CMO came back clear – no obvious cause of OOS DAR
- HIC result comparable when repeated by Sterling, confirming the low DAR value – no obvious cause of OOS DAR
- Prior characterisation knowledge could not pinpoint error – no obvious cause of OOS DAR
- Orthogonal testing showed the OOS batch had a similar oxidation profile to forced degradation – potential cause of OOS DAR
At Sterling, low DAR events have previously been associated with contaminated DMA. Based on this precedent, it was postulated that potential DMA contamination may have occurred, contributing to an oxidative effect that resulted in an OOS low DAR.
DMA is a solvent used in ADC manufacturing, particularly during conjugation, to solubilise hydrophobic drug–linker molecules and maintain their compatibility in aqueous antibody solutions, thereby enabling efficient conjugation.
Root cause investigation – confirm hypothesis
Two third-party DMA lots were assessed alongside an in-house verified DMA used in the previous experiment (see Figure 4) using a scale-down model: Lot 1 (used in the OOS batch) and Lot 2 (a third-party manufacturer alternative).
Following a one-pot reduction, the material was split and treated with the respective DMA lot, then conjugated after 0, 15, or 30 minutes to simulate scale-level processing delays.
Results: The data from this experiment demonstrated that the DMA lot used significantly affects the resulting DAR. DMA quality was identified as the root cause of the OOS low DAR. The 30-minute DMA incubation profile for Lot 1 closely overlays with the OOS drug substance profile (see Figure 5), confirming that solvent quality directly impacted conjugation effciency.
These results highlight the critical influence of DMA quality on the overall success of the conjugation reaction.

Table 3: Process input material Lots – DMA incubation time and corresponding DAR (HIC).
Incubation study
The previous experiment indicated that DMA was the root cause of the OOS failure. To confirm this, an incubation study was performed using DMA and reduced mAb only.
Results: The mAb reoxidised to approximately 50% within 30 minutes and then appeared to plateau (see Figure 6). This behaviour correlates with the drug substance results from the OOS batch, in which the DAR was approximately half of the expected value. This data supports the hypothesis that this lot of DMA promotes reoxidation of the reduced cysteine residues required for conjugation to the linker payload. In the absence of sufficient free thiols, the antibody cannot effciently conjugate to the linker payload, resulting in a reduced DAR.
Although previously not considered a critical process material, this data demonstrates that DMA quality is critical to conjugation success. Prolonged exposure to contaminated DMA induces re-oxidation of the mAb, reducing conjugation efficiency and resulting in lower-than-expected DAR.
DMA impurity analytical development
An investigation was initiated to develop an analytical assay to ensure DMA integrity prior to use. As contaminated DMA material was still available, additional analyses were conducted to determine whether a specific contaminant could be identified. The objective was to enable pre-use assessment of DMA quality, rather than relying on the current proprietary method used in Sterling. A detailed literature review led to the hypothesis that the observed reoxidation of cysteine residues during DMA incubation may be attributed to oxidative degradation products of DMA, which can decompose to form N-methylacetamide (NMA). In-house–verified DMA, third-party manufactured DMA Lot 1 and Lot 2 were analysed by gas chromatography – mass spectrometry (GC–MS) to assess whether a correlation could be established between conjugation performance and the relative abundance of NMA.
GC–MS chromatograms of the three DMA samples (Figure 7) show an impurity eluting at ~3.8 min in the third-party DMA, present at the highest relative abundance and associated with the greatest reduction in DAR (Table 3). The suspected DMA shows ~50% lower impurity levels and a correspondingly smaller DAR reduction, while the in-house verified DMA shows minimal impurity. Collectively, these data indicate that DMA impurity levels correlate with reduced DAR, identifying solvent quality as a key factor affecting conjugation performance.
Conclusion
A lower-than-expected DAR OOS was reported from a batch manufactured at a third party. The investigation identified contaminated DMA as the root cause, particularly when the mAb was incubated for an extended period with DMA. GC-MS analysis of the DMA revealed a peak, whereby its intensity correlated with the extent of reduced DAR. This work highlighted the critical influence of DMA on the overall success of the conjugation reaction.
Further work will focus on:
- Forcing oxidation of DMA to generate the contaminants observed in GC-MS and correlating these results with process data to evaluate their impact onmAb reoxidation and to define the contamination thresholds that lead to mAb reoxidation.
- Transitioning away from Sterling’s proprietary mechanism to determine suitability of DMA and toward physicochemical testing.
- Determining qualified strategies to
- Store DMA and define shelf life
- Reuse availability
- Define approved suppliers/sources
Acknowledgments
The authors would like to thank the following for their assistance with this work:
- Sterling Pharma Solutions’ Deeside process and analytical development and Cramlington teams
- BioPharmaSpec’s Impurity Analysis Services
- Gerardo Zapata and Payam FarahaniADC Therapeutics











